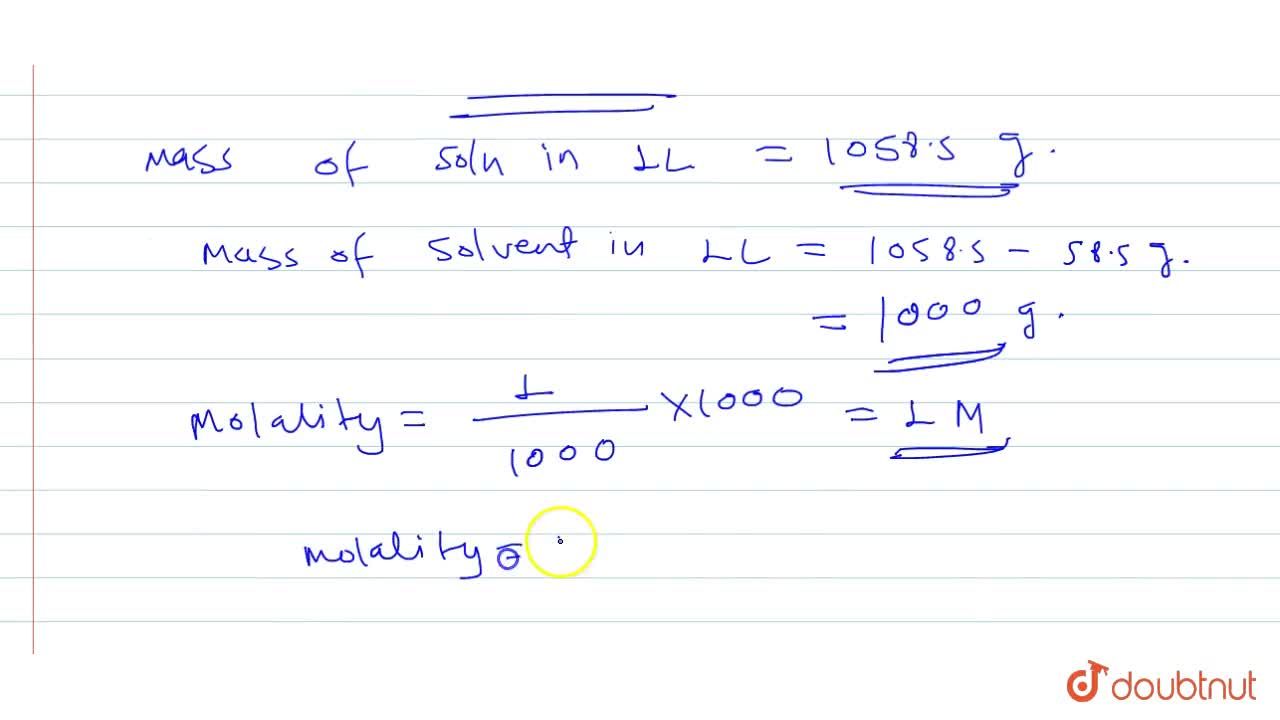1 Molar solution of sodium chloride | 1M solution of sodium chloride | 1 molar solution of NaCl - YouTube

1 Molar solution of sodium chloride | 1M solution of sodium chloride | 1 molar solution of NaCl - YouTube

SOLVED: How would you prepare 1 liter of 250 mM NaCl? and then how would you prepare 0.5 liter of 250 mM NaCl? How would you prepare 1 liter of 173 mM NaCl?

The density of `1 M` solution of `NaCl` is `1.0585 g mL^(-1)`. The molality of the solution is - YouTube

a) rel viscosity of (o) F88, (•) F88 in 1M NaCl, (Δ) P84, (△) P84 in... | Download Scientific Diagram

SOLVED: You have the following stock solutions: 5M NaCl, 1M MgCl2, 2M Tris-HCl. You want to make 50 mL of the following final buffer: 25 mM NaCl, 2.5 mM MgCl2, 0.5M Tris-HCl.
![SOLVED: 1M NaCl solution (solution #1): 1. Mass of NaCl needed to prepare 1O0 mL of 1 M solution 2. Actual mass of NaC] used 853 5.8496 3. Concentration of a solution SOLVED: 1M NaCl solution (solution #1): 1. Mass of NaCl needed to prepare 1O0 mL of 1 M solution 2. Actual mass of NaC] used 853 5.8496 3. Concentration of a solution](https://cdn.numerade.com/ask_images/3b93bfb78214475a88e664f22e1eea93.jpg)
SOLVED: 1M NaCl solution (solution #1): 1. Mass of NaCl needed to prepare 1O0 mL of 1 M solution 2. Actual mass of NaC] used 853 5.8496 3. Concentration of a solution

Consider the following statements. S1 : During electrolysis of 1M NaCl solution Cl 2 does not form at the anode. S2 : For a concentration cell with its reaction at equilibrium both

Cyclic voltammograms acquired on Zn/CPE, in NaCl 1M solution containing... | Download Scientific Diagram